
Diabetes study trial
Class aptent taciti sociosqu ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae commodo. Duis ornare bibendum.

What is Diabetes study trial?
Class aptent taciti sociosqu ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae commodo. Duis ornare bibendum dolor at pellentesque. Proin mattis, tellus ut ultricies facilisis, ante mi consectetur turpis, vel viverra sapien
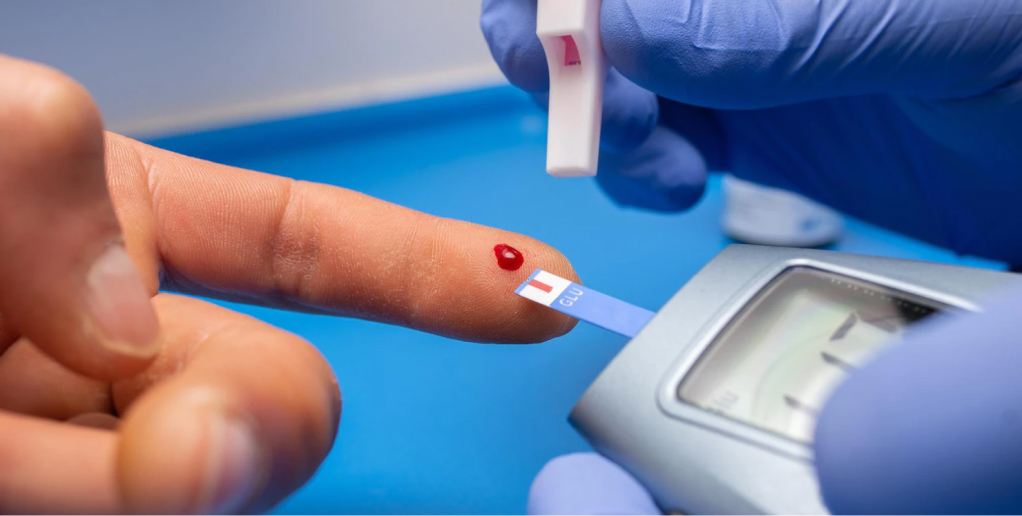
How it works?
Class aptent taciti sociosqu ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae commodo. Duis ornare bibendum dolo
Himenaeos Integer tellus ante, cursus ac sapien sed, varius dictum arcu.
Aptent taciti sociosqu ad litora torquent.
Sed mi dolor, pharetra sit amet

Feature One
Class aptent taciti ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae.

Feature Two
Class aptent taciti ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae.

Feature Three
Class aptent taciti ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae.

Feature Four
Class aptent taciti ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae.

Feature Five
Class aptent taciti ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae.

Feature Six
Class aptent taciti ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis vitae.

Check your eligibility
Take up this questionnaire to help us determine if you have symptoms that are common to this clinical study. if you have the common symptoms, you may be eligible to take part in the study.
Your journey
Class aptent taciti sociosqu ad litora torquent per conubia nostra, per inceptos himenaeos. Duis viverra et felis
Receiving the medication
You would receive entanercept (Enbrel) twice a week for 12 weeks and then once a week for 12 weeks. Etanercept as well as study related medical care is provided at no cost.
Visiting the study site
Study participation involves approximately 8 visits to your local study center over 6 to 7 months.
Follow-up
There would also be a follow-up telephone call 30 days after completing the study. No visits are required after participation is complete.


